The Term Polar Is Used to Describe Molecules Because
The atom with the stronger electron attraction the more electronegative element acquires a slightly negative charge. In molecular terms describe how emulsifiers are able to mix polar and non-polar substances together.
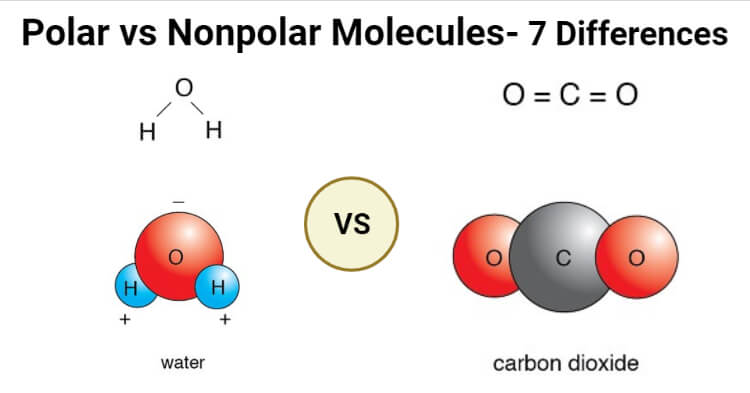
Polar Vs Nonpolar Molecules Definition 7 Key Differences Examples
B polar covalent molecules were first discovered in polar bears.

. Water is polar because of the differences in the electronegativity between oxygen and hydrogen. Polar molecules dissolve easily in water. The charge and high degree of hydration of such molecules prevents them from entering the hydrocarbon phase of the bilayer.
A cell membrane controls what goes in and out of the cell. The term _____ is used to describe a diverse group of non-polar hydrophobic organic molecules. - An emulsifier molecule has two parts the hydrophilic portion and the hydrophobic portion.
Of relating to or being a compound such as a surfactant consisting of molecules having a polar water-soluble group attached to a water-insoluble hydrocarbon chain also. A molecule is polar because it has an uneven distribution of electrons within it. Being a molecule of such a compound.
The single common feature of these molecules is that they are only slightly soluble in water lipid Phospholipids have a _____ polar head and a _____ tail. Now list two molecules that have polar covalent bonds but were nonpolar because the geometry of the molecule causes the pulling to cancel out. By contrast lipid bilayers are highly impermeable to charged molecules ions no matter how small.
When 2 different atoms are joined by a covalent bond and the bonding electrons are shared unequally the bond is a polar covalent bond or POLAR BOND. Small uncharged polar molecules such as water or urea also diffuse across a bilayer albeit much more slowly Figure 11-1. A water molecule is formed when an oxygen atom covalently bonds to two hydrogen atoms.
When dissolved in water the ions _____ and are therefore free to move. Molecules often contain polar bonds because of electronegativity differences but have no overall dipole moment if they are symmetrical this means the overall charges are evenly spread out. Describe in your own words how the overall polarity of a molecule is determined.
A bilayer of phospholipid molecules forms a plasma membrane. When the phospholipid molecules are joined by other lipids and integral proteins the surface can. Because it is a polar molecule.
101 The term polar is used to describe molecules because A polar covalent molecules are found in colder climates. This is an example of polar covalent chemical bonding. The inequality in electron distribution accounts for the bent shape of the molecule.
C there are at least two distinct ends of the molecule regarding electron position and the resulting charge. The membrane lipid molecules are amphipathic. The single common feature of these molecules is that they are only slightly soluble sin water.
For example in the molecule tetrachloromethane CCl 4 the chlorine atoms are more electronegative than the carbon atoms and the electrons are drawn. List two molecules that are nonpolar because the bonds were nonpolar. C there are at least two distinct ends of the molecule regarding electron position and.
B polar covalent molecules were first discovered in polar bears. The term used to describe this differential movement of molecules across a membrane is. Water H 2 O is polar because of the bent shape of the molecule.
The key difference between polar and dipolar molecules is that polar molecules have two opposite ends with opposite electrical charges whereas dipolar molecules have two poles. In brief we can use the terms polar and polar interchangeably because both these terms describe a single molecule having two opposite ends. A phospholipid is an amphiphilic molecule consisting of a polar head region a unit of glycerol and two or more non-polar fatty acid tails typically found in a cell membrane.
For example the oxygen in H2O has a higher electronegativity than the hydrogen. Examples of Molecules with Polar Covalent Bonds. A solid ionic substance does not conduct an electric current because the ions are _____ in place in the crystal structure.
What term is used to describe ions that are closely surrounded by solvent molecules. Oxygen is highly negative compared to hydrogen. The oxygen side of the molecule has a net negative charge while the two.
If a molecule has areas where there is a partial positive or negative charge it is called polar or hydrophilic Greek for water-loving. The term ___ is used to describe a diverse group of non-polar hydrophobic organic molecules. The reason the shape of the molecule isnt linear and nonpolar eg like CO 2 is because of the difference in electronegativity between hydrogen and oxygen.
The less electronegative atom acquires a. Water H 2 O is polar because of the bent shape of the molecule. They act as link between two immiscible substances attracting the polar substance with one end hydrophilic head and the non polar molecules with the other hydrophobic.
The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. The polar glucose molecules are attracted to the. Are phospholipids considered amphipathic.
Because of the hydrophilic and hydrophobic regions of the phospholipid bilayer small nonpolar molecules cross the membrane easily while large and hydrophilic molecules cannot cross easily. The electronegativity value of oxygen is 344 while the electronegativity of hydrogen is 220. A hydrophilic molecule is a term used to describe a molecule that can bond with water meaning that it is attracted to water molecules.
Lipid The two strands of the DNA double helix are held to each other by ___ bonds. The term polar is used to describe molecules because A polar covalent molecules are found in colder climates. This term arises because hydrophobic molecules do not dissolve in water.
The most numerous are the phospholipids. Water H 2 O is a polar bonded molecule.

Mole Definition Teaching Science Chemistry

Why Is Water A Polar Molecule Water Molecule Molecules Polarity Of Water
No comments for "The Term Polar Is Used to Describe Molecules Because"
Post a Comment